By Stem Cells Translational Medicine –
Background
The objective of this study was to assess the long-term effectiveness and safety of implanting mesenchymal stem cells derived from umbilical cord tissue (UC-MSC) in patients diagnosed with retinitis pigmentosa (RP).
Methods
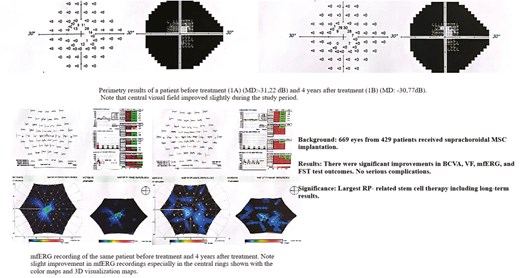
In this single-center study with a retrospective design, 669 eyes received suprachoroidal implantation of 5 million UC-MSCs. Postoperative assessments were conducted on the first day, third month, and every 6 months thereafter. At each visit, evaluations included best-corrected visual acuity (BCVA), anterior segment and fundus examinations, fundus photography, optical coherence tomography, and visual field (VF) tests. Multifocal electroretinography (mfERG) and full-field stimulus threshold (FST) testing were performed at baseline and every 6 months post-therapy. Procedure-related ocular and systemic complications were methodically documented.
Results
A total of 669 eyes from 429 patients underwent surgical intervention. Bilateral procedures were performed in 240 patients, while 189 patients received surgery in only 1 eye. All 669 eyes completed the 12-month follow-up, while 265 eyes completed 2 years, 128 eyes completed 3 years, and 19 eyes completed 4 years of follow-up. No notable ocular or systemic complications were reported during the study duration. Statistically significant improvements in BCVA, VF, and mfERG central rings amplitude measurements were observed over time. FST testing revealed significant improvements in visual sensitivity in 27 patients.
Conclusions
This investigation confirms the long-term benefits and safety profile of suprachoroidal UC-MSC therapy in cases of RP, demonstrating significant improvements in BCVA, VF, mfERG, and FST test outcomes. The data support the feasibility and potential of cell-based therapies as a promising and effective strategy for managing degenerative retinal diseases.